
After the whole solution turns blue, add distilled water. The Hydrochloric Acid turns the Cobalt solution blue. What color does cobalt turn?Ĭhloro Complexes of Cobalt(II) Pour concentrated Hydrochloric acid into the Cobalt Chloride solution. The simplest ion that cobalt forms in solution is the pink hexaaquacobalt(II) ion – 2 +. However cobalt does not react with water that is at room temperature. Does cobalt react with water?Ĭhemical Properties Cobalt has the ability to react with most acids to produce hydrogen gas. Its use requires appropriate precautions to avoid internal contamination and to prevent cobalt poisoning. Is cobalt blue poisonous?Ĭobalt blue is toxic when ingested or inhaled. Only 18 cases have been reported, 28, 30 – 37 ten involving MoM articulations. The development of systemic cobalt toxicity from hip arthroplasty appears to be very rare. 
Elemental it occurs only very rarely in meteorites and in the earth’s core. This puts it in the thirtieth place in the list of frequency ordered items. 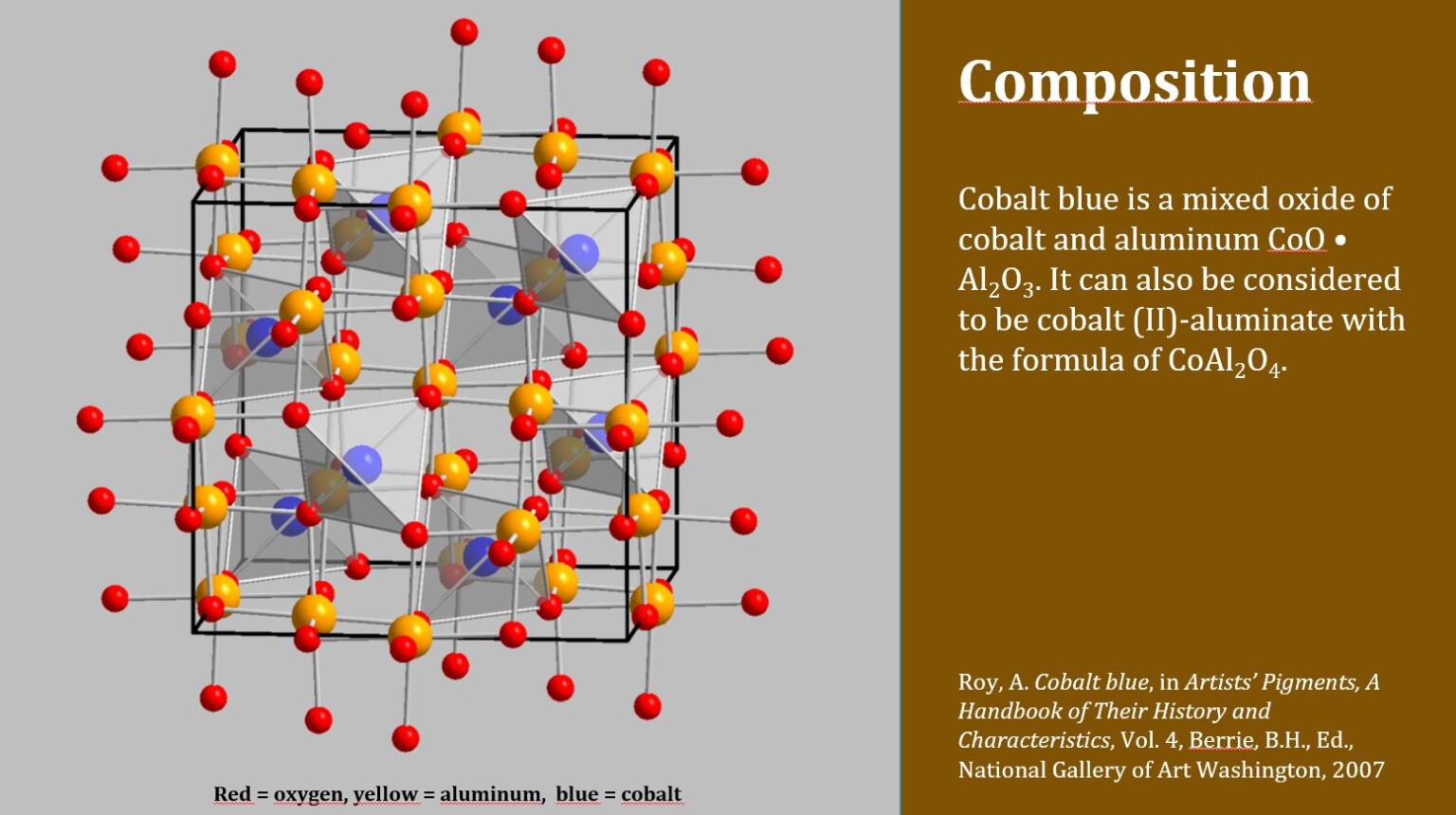
Rare Cobalt is a rare element with a frequency in the Earth’s crust of 0,004 percent. Inductively coupled plasma/mass spectrometry (ICP/MS): An argon plasma at 6,000-10,000 K destroys the organic matter in the sample and ionizes the metals. How do you test for cobalt?ġ,2 Laboratory testing is needed to detect high levels of cobalt in the body that may lead to cobalt toxicity. Only ferromagnetic materials such as iron, cobalt, and nickel are attracted to magnetic fields strongly enough to be truly considered magnetic. As the total global cobalt reserves amount to 7.6 million metric tons, this means that the DR Congo’s cobalt reserves account for nearly half of the world’s reserves of the metal. The Democratic Republic of the Congo has the largest cobalt reserves in the world, at some 3.5 million metric tons as of 2021. Cobalt can be used to make both soft and hard magnets. Over the past 100 years, cobalt has been used widely because of its excellent magnetic properties. Cobalt-60 is a commercially important radioisotope, used as a radioactive tracer and for the production of high-energy gamma rays. Is raw cobalt radioactive?Ĭobalt occurs naturally as only one stable isotope, cobalt-59. Workers may be harmed from exposure to cobalt and cobalt-containing products. It can harm the eyes, skin, heart, and lungs. 1735) by Swedish chemist Georg Brandt, though cobalt compounds had been used for centuries to impart a blue colour to glazes and ceramics. What metal is naturally blue?Ĭobalt (Co), chemical element, ferromagnetic metal of Group 9 (VIIIb) of the periodic table, used especially for heat-resistant and magnetic alloys. This is because the chlorine ions force the water out of the water complex of cobalt. Adding concentrated hydrochloric acid to the solution changes the color of the solution to bluish-purple. Pink color Cobalt ions are hydrated in the solution and have a pink color. Aside from some notable spikes it tends to trade around $15 USD per pound. How much is cobalt worth?Ĭobalt is most commonly used in alloys and batteries. It was traditionally used to colour glass and ceramics. The pigment that was traditionally extracted from a mineral comes, however, in various shades, varying from yellow to red and from blue to green. In the art of painting cobalt is mainly known as a colour blue. It is similar to iron and nickel, according to the Environmental Protection Agency, and - like iron - can be made magnetic. In pure form, cobalt is silvery-blue and brittle. Cobalt is predominantly used for producing brilliant blue pigments. It makes a strong alloy with other metals, has a high melting point and low conductivity. As a pure metal, cobalt is hard and brittle. What does the mineral cobalt look like?Ĭobalt is a grayish-white metal with the chemical symbol Co. Cobalt can also be found in soil and crusts on the seafloor. Can pure cobalt be found in nature?ġ) Pure Cobalt Cannot be Found in Nature In fact, cobalt has long been considered a by-product of nickel and copper mining operations. While in it’s natural, raw state it’s a somewhat burnished silver colour, it is famously known for producing vibrant blues in pigments. Silver colour Cobalt, is symbol Co on the periodic table with an atomic weight of 27. Classified as a transition metal, Cobalt is a solid at room temperature. Cobalt is a chemical element with symbol Co and atomic number 27.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
